We want to make carbon-free electrical power accessible to all
The Strontium Alternative Magnet Innovation Consortium (SrAMIC) will investigate the potential for responsibly sourced circular supply chains for high strength strontium ferrite magnets (also known as ceramic magnets) as an alternative to, or to complement, the rare earth permanent magnets used in electric traction motors and in renewable power generation.
Our Mission
Traditional electric motors using rare earth elements are expensive, vulnerable to supply chain risks, and have high environmental impact from mining including radioactive residues. New motor technologies using alternative magnets such as ferrite magnets are more environmentally friendly, abundant, and cheaper to source and manufacture.
Innovations in motor design such as newly modelled structures are helping to close the performance gap between the incumbent rare earth motors and new ferrite magnet alternatives. To achieve comparable efficiency there are trade-offs in specific applications. However, these advancements are highlighting a new dawn for more sustainable and cost-effective motor and generator solutions.
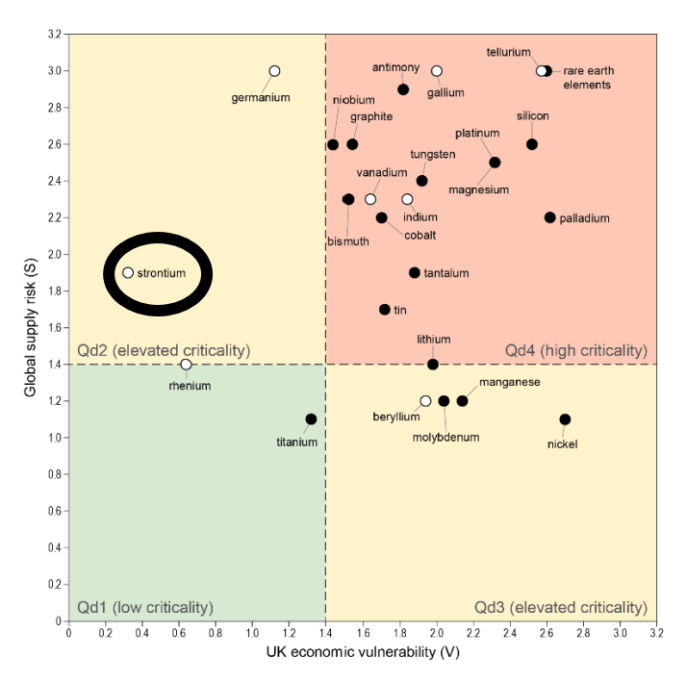
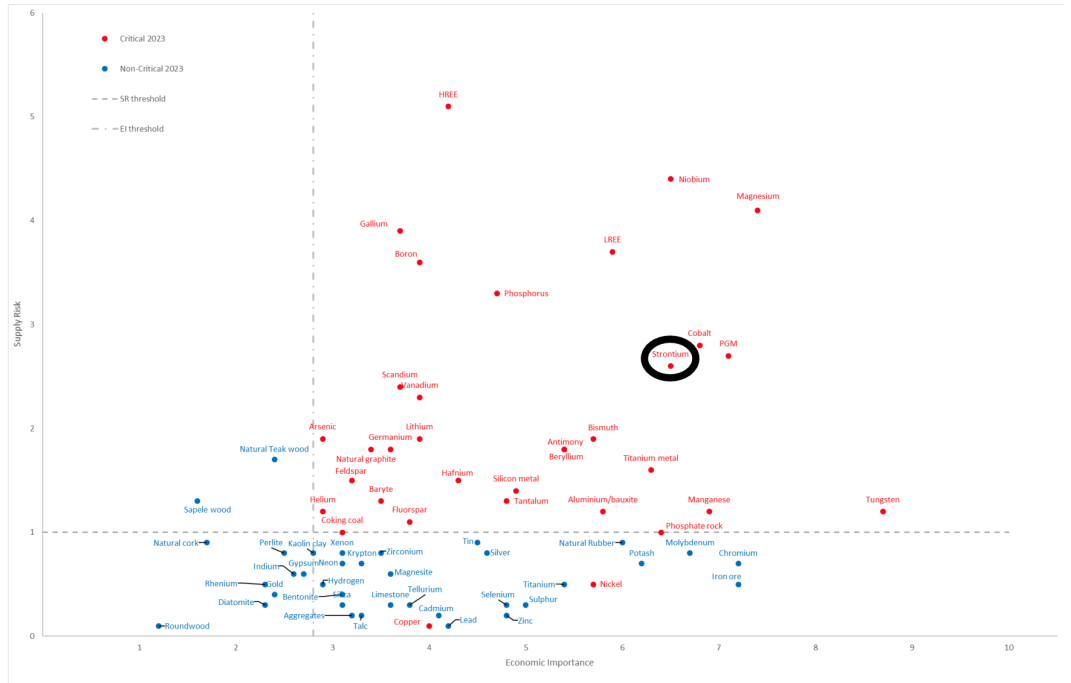
Even though raw materials for ferrite manufacturing are more abundant and accessible than those for NdFeB magnets, strontium (a vital component for higher grade ferrites) is starting to appear on some critical minerals lists. Strontium is currently sourced from the mineral celestine, but its conversion from strontium sulphate to strontium carbonate (for direct use in ferrite) is a polluting and energy intensive process. SrAMIC are investigating bypassing the need to convert strontium sulphate in the production of ferrite magnets.
EU position: strontium on par with cobalt and PGMs
Current processing Techniques
Today celestite is converted in strontium carbonate using either the Black Ash process or Soda Ash process. Both of these processes are carbon emitting, energy intensive, and reagent intensive. SrAMIC is aiming to change this.
Current black Ash Process:
- Finely ground celestite is mixed with coal and heated in a rotary kiln at high temperatures (around 1300°C).
- The resulting strontium sulfide is then leached with water to form a solution of strontium hydroxide and other impurities.
- Carbon dioxide is bubbled through the strontium hydroxide solution, precipitating strontium carbonate out of the solution.
- The strontium carbonate is filtered out, washed to remove impurities, and then dried to obtain the final product.

Current Soda Ash Process:
- Finely powdered celestite is mixed with soda ash (sodium carbonate).
- The mixture is treated with steam for one to three hours. During this time, a chemical reaction occurs, converting the celestite into strontium carbonate and sodium sulfate.
- The resulting mixture contains less soluble strontium carbonate and more soluble sodium sulfate. These are separated by centrifugation.
- The strontium carbonate is filtered out, washed to remove impurities, and then dried to obtain the final product.
- The soda ash route struggles to obtain ferrite grade strontium carbonate.

We need your consent to load the translations
We use a third-party service to translate the website content that may collect data about your activity. Please review the details in the privacy policy and accept the service to view the translations.